|
What do others say?Īlthough AstraZeneca has been in favor of spacing out shots, Moderna and Pfizer have been more cautious. The World Health Organization says that, although there is some data to support that interval for the AstraZeneca vaccine, there is no scientific evidence to stretch the second dose of the Pfizer-BioNTech vaccine to 12 weeks. drugs regulator has recommended the AstraZeneca vaccine be administered within an interval of up to 12 weeks. Data collected by AstraZeneca, however, shows that pacing out doses by 8 to 12 weeks may actually be the efficacy “sweet spot,” Mene Pangalos, the drugmaker’s executive vice president for biopharmaceutical research and development, told a U.K. However, some data collected from older residents vulnerable to developing severe illness indicated the first dose led to a 33% reduction in COVID-19 cases, far lower than the effectiveness after two doses. Health officials there have noted that cases started to decline within two weeks of the shots being administered, and before people began receiving a second dose. What’s happened in countries with rapid vaccination?īy late January, Israel had given more than a quarter of its people at least one dose. And the social risk is that mixed messages on the topic-from governments, regulators, professional groups and drugmakers-will make it more difficult to communicate with the public on vaccines that are already controversial, and shunned by some. This would undermine a year’s work to develop safe and effective vaccines. The theoretical risk is that a weak response to the virus in millions of people who have received just one dose would foster the emergence of new variants that could evade vaccine-induced antibodies. Spacing out doses extends the period when people-most of whom have been chosen to get vaccines quickly at this point for reasons of vulnerability-will have incomplete protection until they get their second dose. 
The medical risk is that the first dose almost certainly won’t be as protective as two doses. In a split with other regulators, the European Medicines Agency said that changing how coronavirus vaccines are given to people raises the risk of reducing effectiveness.ĥ. There is also concern that inoculated patients may change their behavior in a way that increases the risk of spread, negating some of the benefits of vaccination. There are no data to demonstrate that protection after the first dose is sustained after 21 days. Data from the Pfizer-BioNTech trial found partial protection was achieved as early as 12 days after the first dose, but two doses were required to provide maximum efficacy, or 95% protection against disease. 
The FDA said slight delays shouldn’t affect the protection offered by the vaccine. 
The new guidance said doses can be spaced out by up to six weeks if it’s impossible to get the follow-up shot on time. health authorities to revise guidance that recommended an interval of three or four weeks between doses of the Moderna and Pfizer-BioNTech vaccines. aims to give more people an initial dose. What’s being proposed?īy using shots that normally would have gone to people who have already received the first dose, the U.K. Johnson & Johnson is studying a single-shot COVID-19 vaccine, with results expected to be analyzed soon. COVID-19 vaccines may work the same way, but it will take additional studies to know this for sure. Some vaccines requiring multiple doses are administered four months apart to optimize immune memory. Typically booster shots are intended to provide the immune system with advanced “training” to make better antibodies. Vaccines are intended to hardwire immunological memory to generate a rapid, targeted and durable response by antibodies and T-cells to an invading foe. Doses of AstraZeneca’s vaccine were scheduled 4 to 12 weeks apart across four trials. In studies, the second Pfizer-BioNTech dose was given 21 days after the initial shot Moderna’s was given 28 days later. The recommended timing of the second dose varies depending on the vaccine, and is based on clinical trial data regulators used in their review process. 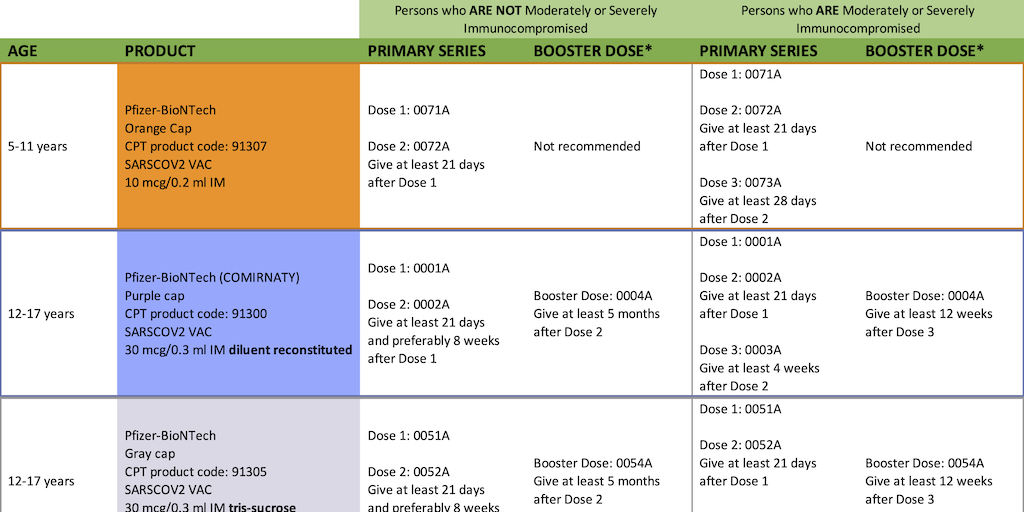
COVID-19 vaccines made by Moderna Inc., Pfizer Inc.-BioNTech SE, and AstraZeneca Plc are administered in two doses.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
